Summary Card
Overview
There are 4 main groups: acids, burns, organic solvents, and inorganic solvents.
Features
The degree of chemical burn is dependent on the chemical, its concentration, and the extent of exposure.
Complications
Each chemical has specific secondary effects. For example, hydrofluoric acid has severe pain, low calcium, and raised potassium.
Management
Treat with prompt & constant water irrigation (some exceptions!). After neutralisation, manage similar to a thermal burn.
Overview of Chemical Burns
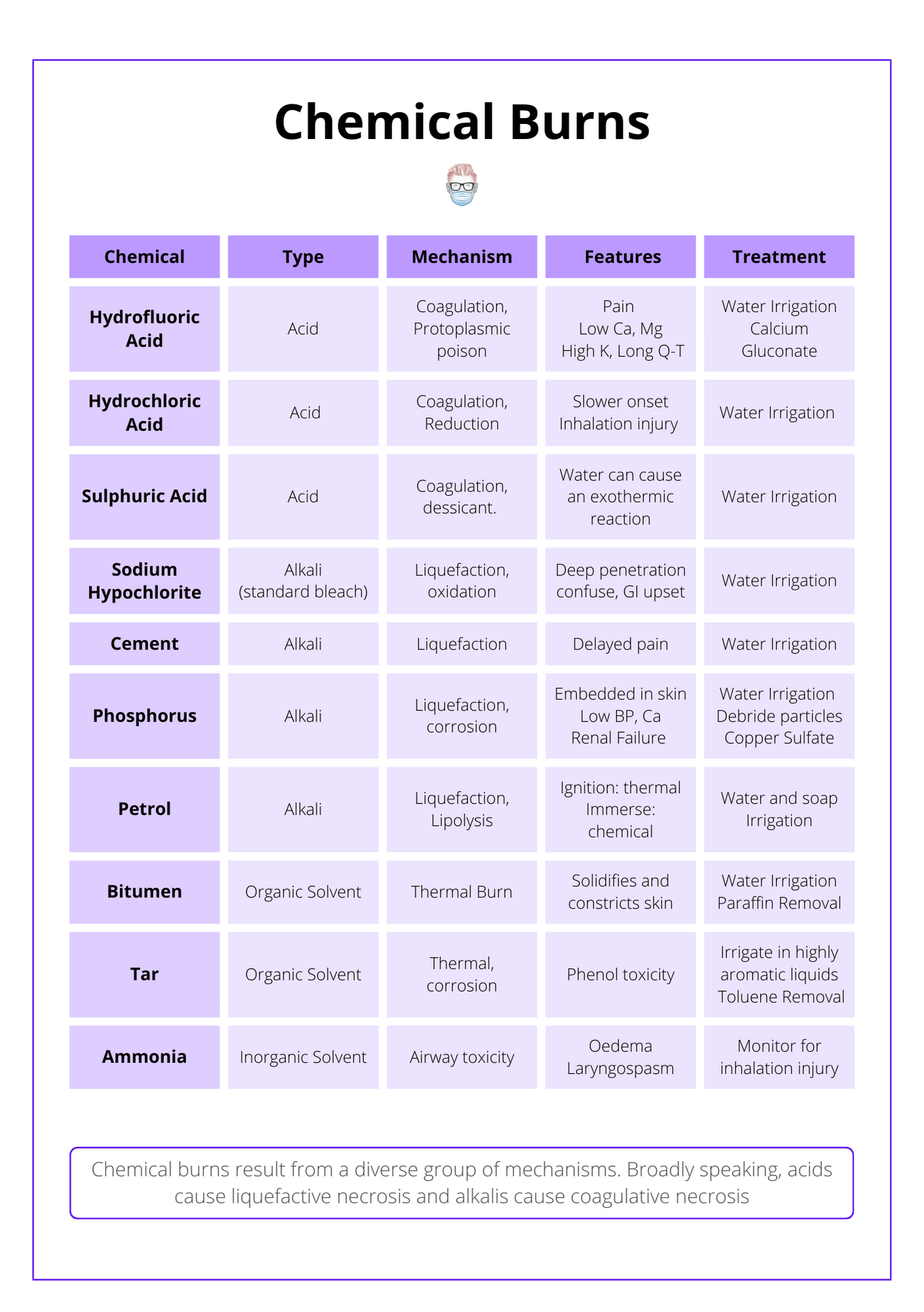
The 4 main groups are acids, burns, organic solvents, and inorganic solvents.
Chemical burns can be broadly categorised into 4 main groups: acids, burns, organic solvents, and inorganic solvents. They differ in their pathophysiology, clinical progression, and potential complications.
Chemical burns are categorised in the table below.
Features of Chemical Burns
The degree of a chemical burn is dependent on the type of chemical, the extent of exposure, and first aid management. Each chemical has a specific set of secondary features or complications.
The degree of a chemical burn can vary based on the type of chemical, the extent of exposure, and any first aid management.
Degree of Burn
The degree of chemical burn is dependent upon the following variables:
- Type and extent of exposure (skin, mucosa, inhalation)
- Type of chemical agent and its mechanism of action
- Quantity, strength, or concentration of agent
The image below illustrates chemical burns on skin.
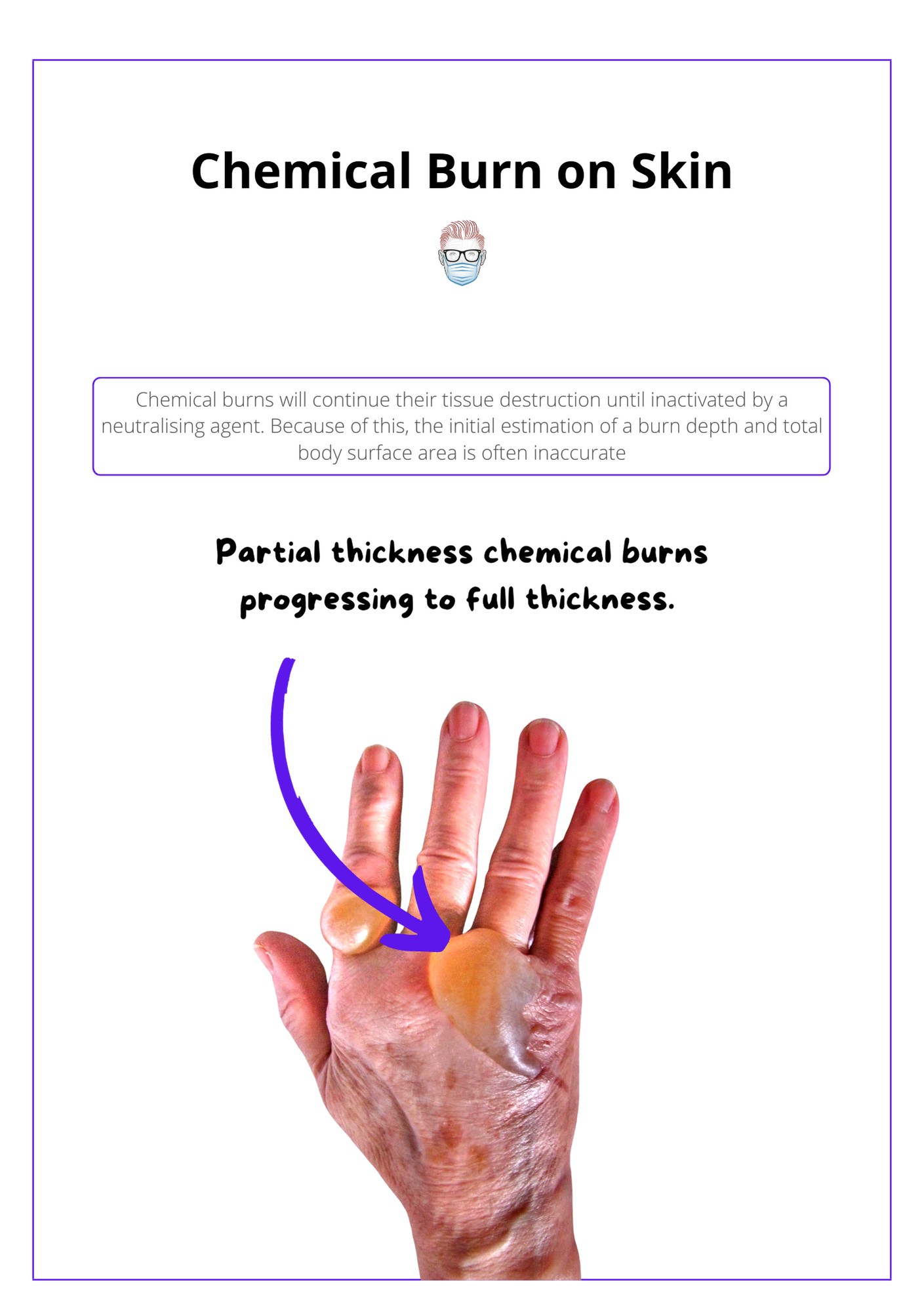
Unlike thermal burns, chemical burns will continue their tissue destruction until inactivated by a neutralising agent. Because of this, the initial estimation of a burn depth and total body surface area is often inaccurate.
Complications of Chemical Burns
Each chemical has specific secondary effects. For example, hydrofluoric acid has severe pain, low calcium, and raised potassium.
Each chemical has its own mechanism of burn. As a result, each chemical can often have a specific set of secondary clinical presentations. This can help guide the type of chemical involved and can also help treat the patient.
Below is a visual summary of complications of chemical burns.
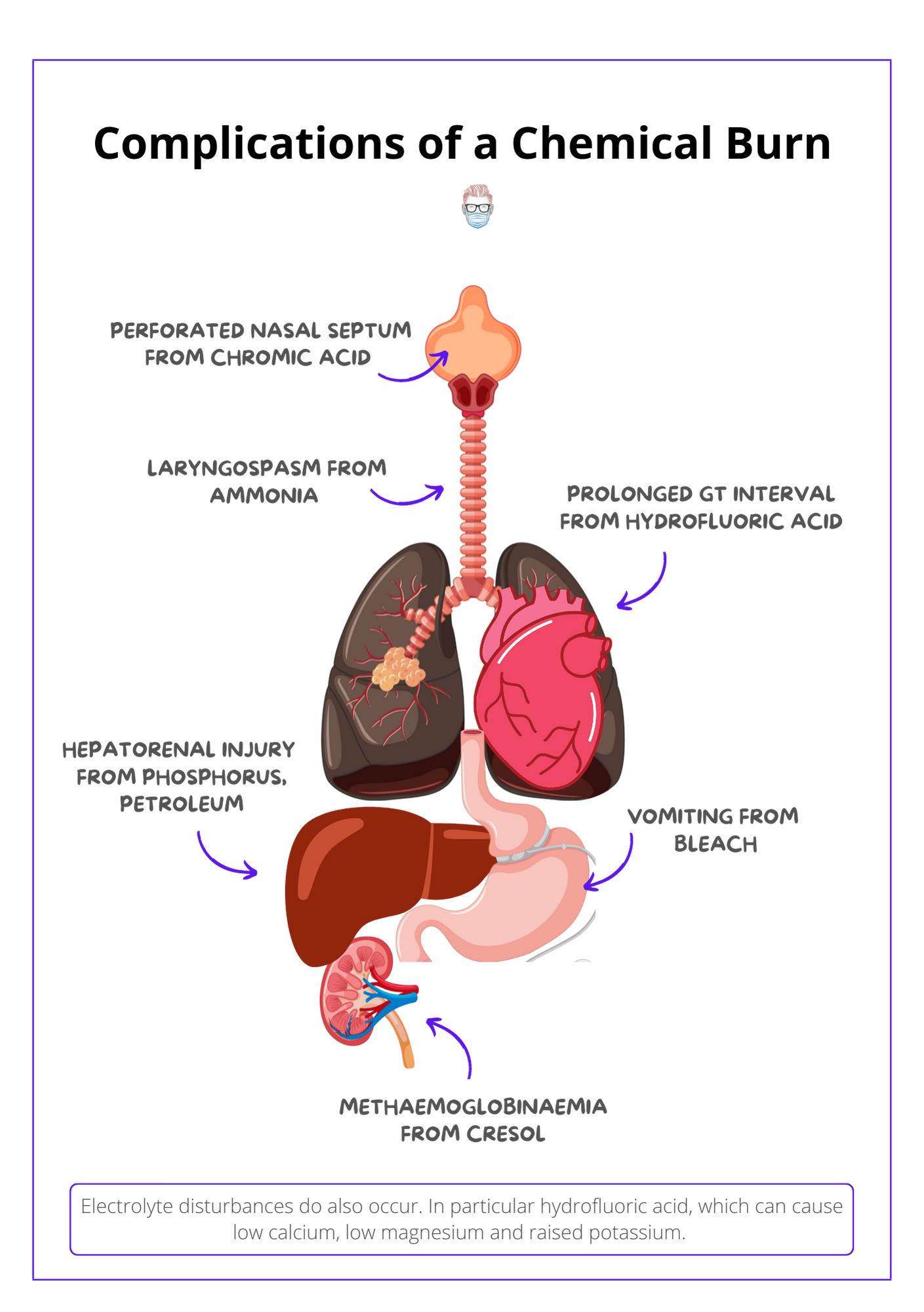
- Acute pain: hydrofluoric acid
- Hypocalcaemia: hydrofluoric acid, phosphorous
- Hypomagnesaemia: hydrofluoric acid
- Hyperkalaemia: hydrofluoric acid
- Prolonged Q-T Interval: hydrofluoric acid
- Hepatorenal injury: tannic, formic acid, phosphorous, petroleum
- Particles embedded in the skin: phosphorus
- Inhalation injuries: ammonia
- Methaemoglobinaemia and hemolysis: cresol
- Perforated nasal septum: chromic acid
Management of Chemical Burns
Chemical burns are typically treated with prompt and constant water irrigation (some exceptions!). After neutralisation of the chemical, treat complications and manage similar to a thermal burn.
Chemical burns are generally treated with prompt and constant water irrigation with some exceptions.
General Management
- Remove causative agent and any contaminated clothing.
- Constant water irrigation whilst monitoring for pH.
- Monitor systemic toxicity or complications.
- Assess for specific injuries: eyes, airway, hair, nails, heart, kidneys.
- Discuss or contact toxicology experts.
- Once neutralised, treat similarly to a thermal burn.
There are specific exceptions to the "constant irrigation with water". These are:
- Sodium, potassium, and lithium may ignite.
- Phenol is not water-soluble (apply polyethylene glycol).
- Hydrochloric acid + H20 may cause exothermic reactions (neutralise by soap).
- Diphoterine binds to acids, alkalis, vesicants, and many more substances.
Hydrofluoric Acid
In addition to the above management, hydrofluoric acid requires neutralisation with calcium gluconate to inactivate free fluoride ions.
- Topical calcium gluconate burn gel (10% with DMSO).
- Injection with 10% calcium gluconate (multiple 0.1–0.2 ml via 30G needle).
- Intra-arterial infusion of calcium gluconate.
- Intravenous ischaemic retrograde infusion (Biers block).
- Early excision is sometimes required.
Secondary Injuries
- GI Tract: endoscopy and CT may show extra-luminal damage.
- Lungs: fibre-optic bronchoscopy, bronchodilators.
- Eyes: ophthalmology referral.
Conclusion
1. Chemical Burn Categories: You've gained insight into the four main categories of chemical burns, including acids, burns, organic solvents, and inorganic solvents, and how they differ in their pathophysiology and clinical progression.
2. Burn Severity Factors: You now understand how the type of chemical, its concentration, and the duration of exposure influence the severity of chemical burns and their initial management.
3. Identifying Complications: You've learned about specific secondary effects and complications associated with different chemicals, crucial for effective emergency response and treatment planning.
4. Management Techniques: You are now familiar with the initial management of chemical burns, including the importance of prompt and constant water irrigation and the exceptions where different treatments are required.
5. Treatment Protocols: You've explored detailed protocols for managing complex cases and the necessary steps for addressing systemic and local injuries caused by chemical exposures.